Principal Investigator
Peter Vandenabeele
Research interests
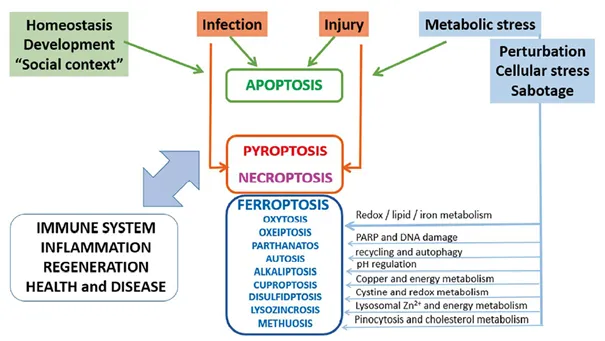
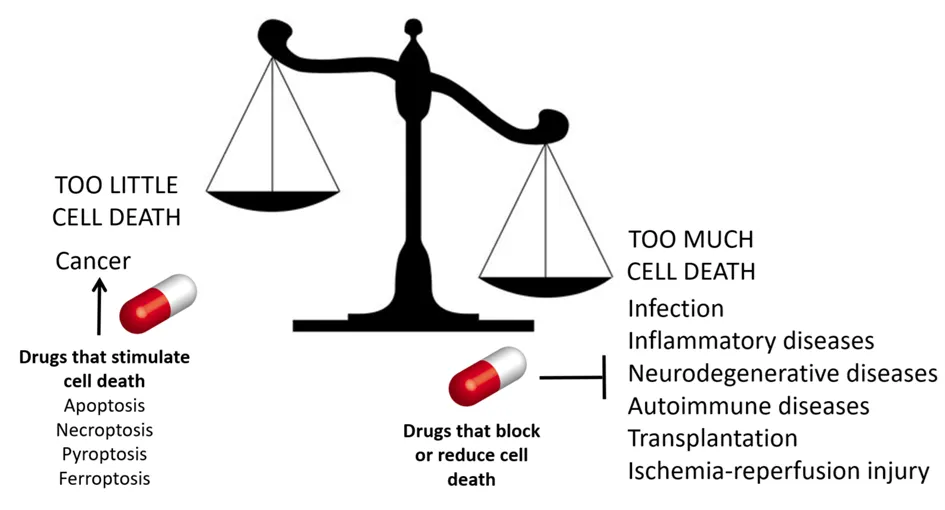
Cell death is essential for maintaining homeostasis. Under healthy, steady-state conditions, approximately 100 billion (10¹¹) cells die each day—equivalent to about one million cells per second. In pathological contexts, however, cell death becomes dysregulated: excessive cell death contributes to inflammatory and degenerative diseases, whereas insufficient cell death is a hallmark of cancer.
The Cell Death and Inflammation Unit brings together four research teams that address interconnected aspects of cell death and its impact on inflammation, cancer, infection, and immune regulation (Vandenabeele Team, Bertrand Team, Maelfait Team, and Declercq Team). By combining complementary expertise, the Unit aims to generate fundamental insights into cell death mechanisms and translate these findings into therapeutic strategies.
The Vandenabeele Team focuses on immunogenic cell death and cancer immunotherapy, with particular emphasis on enhancing the immunogenicity of ferroptosis to improve cancer treatment outcomes. Ferroptosis is a regulated form of cell death driven by lipid peroxidation (LPO) resulting from disturbances in iron, redox, and lipid metabolism. One of our recent key findings is that ferroptotic dying cells produce immunosuppressive signals that interfere with antigen cross-presentation preventing an adequate antitumor response. By elucidating the underlying mechanisms—particularly those involving lipid droplet formation—we aim to develop strategies to increase the immunogenicity of ferroptotic and irradiated cancer cells, two processes that share lipid peroxidation as a common feature.
Selected publications
- Wiernicki B, Maschalidi S, Pinney J, Adjemian S, Vanden Berghe T, Ravichandran KS, Vandenabeele P. Cancer cells dying from ferroptosis impede dendritic cell-mediated anti-tumor immunity. Nat Commun. 2022 Jun 27;13(1):3676. doi: 10.1038/s41467-022-31218-2. PMID: 35760796
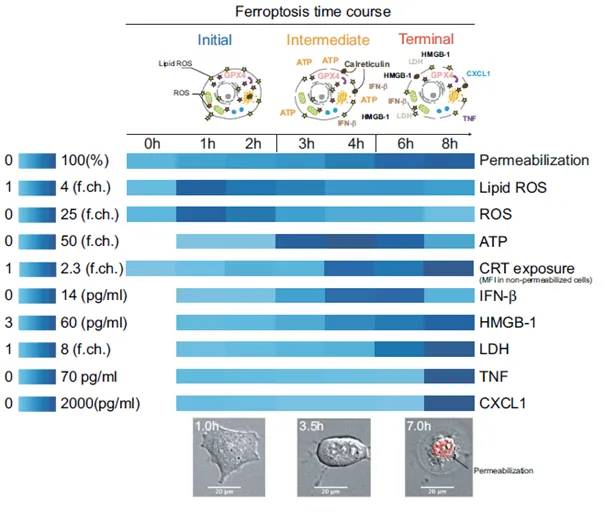
- Our study defines three stages of ferroptosis (initial, intermediate, terminal) and demonstrates that early ferroptotic cancer cells impair dendritic cell (DC) maturation, uptake, and antigen cross-presentation.
- DCs exposed to ferroptotic cancer cells fail to generate effective anti-tumor immunity, in contrast to DCs loaded with necroptotic cells.
- Overall, ferroptosis suppresses antigen-presenting cell function and adaptive immune responses, which may limit its therapeutic applicability in cancer treatment.
- Ghazavi F, Huysentruyt J, De Coninck J, Kourula S, Martens S, Hassannia B, Wartewig T, Divert T, Roelandt R, Popper B, Hiergeist A, Tougaard P, Vanden Berghe T, Joossens M, Berx G, Takahashi N, Wahida A, Vandenabeele P. Executioner caspases 3 and 7 are dispensable for intestinal epithelium turnover and homeostasis at steady state. Proc Natl Acad Sci U S A. 2022 Feb 8;119(6):e2024508119. doi: 10.1073/pnas.2024508119. PMID: 35105800
- Although apoptosis has long been considered essential for intestinal epithelial cell (IEC) turnover and immune homeostasis, IEC-specific deletion of caspase-3 and caspase-7 does not affect intestinal architecture, cellular composition, or proliferation.
- The absence of apoptotic signaling is compensated by increased non-apoptotic cell extrusion, thereby preserving normal physiological levels of IEC shedding.
- Microbiome diversity and composition remain unchanged in apoptosis-deficient mice, with no evidence of dysbiosis.
- Transcriptomic and single-cell RNA sequencing analyses show no significant alterations in epithelial differentiation, inflammatory signaling, or immune cell composition, demonstrating that apoptosis is dispensable for intestinal homeostasis under steady-state conditions.
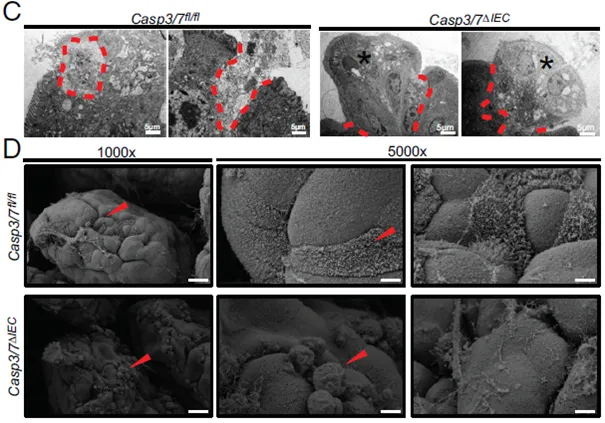
- Xie W, Wyckaert L, Vadi M, Verstraeten B, Divert T, Haerinck J, De Rycke R, Baeke F, Lamkanfi M, Berx G, Wahida A, Vandenabeele P. Caspase-3/7 deficiency results in enhanced intestinal inflammation and reduced tumorigenesis. Sci Adv. 2026 Mar 20;12(12):eadz5906. doi: 10.1126/sciadv.adz5906. Epub 2026 Mar 20. PMID: 41861024
- Among major intestinal epithelial cell (IEC) death pathways, caspase‑3/7–mediated apoptosis plays a uniquely protective role during intestinal inflammation.
- In the DSS colitis model, IEC-specific loss of caspase‑3/7 results in more severe disease, increased mortality, impaired epithelial regeneration, and compromised barrier integrity—phenotypes not observed when necroptosis, pyroptosis, or ferroptosis pathways are disrupted.
- Caspase‑3/7 deficiency suppresses intestinal stem cell proliferation and promotes inflammatory cell death, thereby delaying mucosal repair despite elevated inflammatory responses.
- Notably, although colitis severity is increased, caspase‑3/7–deficient mice develop fewer tumors in colitis-associated colorectal cancer models, indicating a dual role for caspase‑3/7 in limiting inflammation while promoting tumorigenesis following intestinal injury.
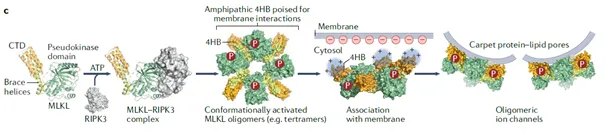
- Vandenabeele P, Bultynck G, Savvides SN. Pore-forming proteins as drivers of membrane permeabilization in cell death pathways. Nat Rev Mol Cell Biol. 2023 May;24(5):312-333. doi: 10.1038/s41580-022-00564-w. Epub 2022 Dec 21. PMID: 36543934
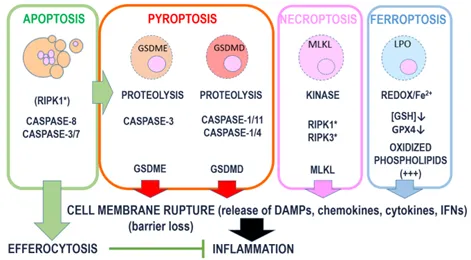
- Regulated cell death (RCD) is executed by pore-forming proteins (PFPs), including BAX, BAK, and BOK in apoptosis, gasdermins in pyroptosis, and MLKL in necroptosis.
- These PFPs are synthesized as inactive precursors and undergo activation through coordinated processes such as proteolytic cleavage or phosphorylation, followed by membrane recruitment, insertion, and oligomerization mediated by protein–protein and protein–lipid interactions.
- The review outlines the structural rearrangements that enable PFPs to transition from autoinhibited states to membrane-embedded pore-forming complexes, resulting in distinct pore architectures.
- It further compares shared and pathway-specific mechanisms among BAX/BAK/BOK, gasdermins, and MLKL, and discusses how the coexistence of multiple cell death modalities within cell populations may have important physiological and pathological consequences.
- Vandenabeele P, Conrad M, Wahida A. For Better or Worse: The Impact of Necrotic Cell Death Modalities. Adv Exp Med Biol. 2025;1481:241-292. doi: 10.1007/978-3-031-92785-0_8. PMID: 41004085
- Diverse cellular stresses, including infection and inflammation, trigger distinct forms of regulated cell death such as apoptosis, necroptosis, pyroptosis, and the metabolically driven process of ferroptosis, depending on the stimulus and cellular context.
- Each cell death modality is associated with a specific membrane‑permeabilizing mechanism, including gasdermin cleavage in pyroptosis and secondary necrosis, MLKL activation in necroptosis, and lipid peroxidation–induced plasma membrane damage in ferroptosis.
- In addition to pore formation, the membrane protein NINJ1 senses cellular swelling (oncosis) and induces catastrophic plasma membrane rupture during the terminal phase of cell death, leading to the release of intracellular contents.
- Elucidating the signaling and metabolic mechanisms underlying regulated necrotic cell death provides therapeutic opportunities to modulate these processes in diseases such as cancer, ischemia–reperfusion injury, inflammation, and degenerative disorders affecting the brain, intestine, and skin.