Principal Investigator
Tom Vanden Berghe
Research interests
Tom Vanden Berghe leads a multidisciplinary research team studying the molecular mechanisms and therapeutic targeting of regulated cell death, with a particular focus on ferroptosis—an iron-dependent form of cell death increasingly implicated in cancer and inflammatory diseases. The laboratory aims to translate fundamental insights in cell death biology into clinically relevant diagnostic and therapeutic strategies.
The research program integrates basic discovery science with translational development. The team investigates the molecular regulation of ferroptosis and related inflammatory cell death pathways and their roles in disease progression, treatment response, and therapy resistance. Based on these insights, the group has established a ferroptosis research platform focused on identifying novel regulators and inhibitory compounds, developing biomarkers and diagnostic approaches, and exploring innovative therapeutic strategies such as targeted nanomedicine and antibody–drug conjugates.
Using integrated experimental systems ranging from molecular and cellular models to advanced preclinical disease models, the laboratory validates mechanistic hypotheses and evaluates therapeutic interventions. In collaboration with clinical partners, the group also studies cell death signatures in human disease, including prospective studies in critically ill patients. These efforts aim to define molecular fingerprints of cell death that can support precision medicine and guide patient stratification and treatment decisions.
Selected publications
- Veeckmans, G., L. Devos, N. Gilbo, D. Van Beersel, C. Scarpellini, J. Blondeel, M. Walravens, G. Klejborowska, C. Lanthier, M. Wölk, S. Müller, C. Gaillet, L. Colombeau, B. Hassannia, M. Längin, M. Bender, J. Abicht, B. Reichart, H. De Winter, M. Fedorova, R. Rodriguez, J. Pirenne, L. J. Ceulemans, I. Jochmans, K. Augustyns, D. Monbaliu, A. Neyrinck, T. Vanden Berghe. Ferroptosis Inhibition Enhances Liver and Lung Graft Function. Cell Accepted (IF 46)
- Van San, E., A.C. Debruyne, G. Veeckmans, Y.Y. Tyurina, V.A. Tyurin, H. Zheng, S.M. Choi, K. Augustyns, G. van Loo, B. Michalke, V. Venkataramani, S. Toyokuni, H. Bayir, P. Vandenabeele, B. Hassannia, and T. Vanden Berghe. 2023. Ferroptosis contributes to multiple sclerosis and its pharmacological targeting suppresses experimental disease progression. Cell Death Differ. 30:2092–2103. doi:10.1038/s41418-023-01195-0. (IF 15, 91 citations)
- Ferroptosis contributes to disease progression in multiple sclerosis (MS).
- MS lesions and cerebrospinal fluid show increased labile iron, lipid peroxidation, and ferroptosis-associated lipid degradation products.
- The ferroptosis inhibitor UAMC-3203 delays relapse and ameliorates disease progression in a preclinical model of relapsing-remitting MS.
- Targeting ferroptosis represents a novel therapeutic strategy that could complement current immunosuppressive treatments for MS.
- Van Coillie, V., E. Van San, I. Goetschalckx, B. Wiernicki, B. Mukhopadhyay, W. Tonnus, S.M. Choi, R. Roelandt, C. Dumitrascu, L. Lamberts, G. Dams, W. Weyts, J. Huysentruyt, B. Hassannia, I. Ingold, S. Lele, E. Meyer, M. Berg, R. Seurinck, Y. Saeys, A. Vermeulen, A.L.N. van Nuijs, M. Conrad, A. Linkermann, M. Rajapurkar, P. Vandenabeele, E. Hoste, K. Augustyns, and T. Vanden Berghe. 2022. Targeting ferroptosis protects against experimental (multi)organ dysfunction and death. Nat Commun. 13:1046. (IF15, 156 citations)
- Plasma catalytic iron and lipid peroxidation correlate with severity of multiorgan dysfunction and mortality in ICU patients.
- Experimental models demonstrate that iron-induced multiorgan dysfunction is mediated by ferroptosis.
- Pharmacological inhibition of lipid peroxidation with a soluble ferrostatin analogue protects against non-septic multiorgan dysfunction in mice.
- Targeting ferroptosis may represent a therapeutic strategy for a stratifiable subset of patients with multiorgan dysfunction syndrome (MODS).
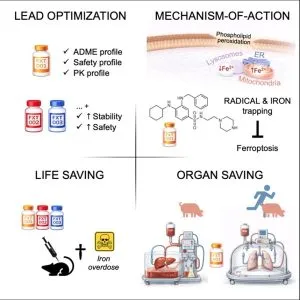
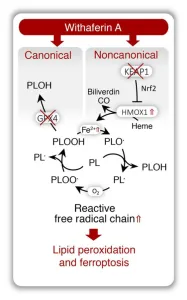
- Hassannia, B., B. Wiernicki, I. Ingold, F. Qu, S. Van Herck, Y.Y. Tyurina, H. Bayir, B.A. Abhari, J.P.F. Angeli, S.M. Choi, E. Meul, K. Heyninck, K. Declerck, C.S. Chirumamilla, M. Lahtela-Kakkonen, G. Van Camp, D.V. Krysko, P.G. Ekert, S. Fulda, B.G. De Geest, M. Conrad, V.E. Kagan, W. Vanden Berghe, P. Vandenabeele, and T. Vanden Berghe. 2018. Nano-targeted induction of dual ferroptotic mechanisms eradicates high-risk neuroblastoma. J Clin Invest. 128:3341-3355. (IF 13, 656 citations)
- Withaferin A induces ferroptosis in high-risk neuroblastoma through a dual mechanism involving GPX4 inhibition and NRF2/KEAP1 pathway activation.
- Excessive HO-1 activation increases intracellular labile Fe(II), driving noncanonical ferroptosis.
- Withaferin A shows superior antitumor efficacy compared with etoposide and cisplatin in neuroblastoma models.
- Nanoparticle-mediated delivery enhances tumor targeting and suppresses neuroblastoma growth and relapse in vivo.
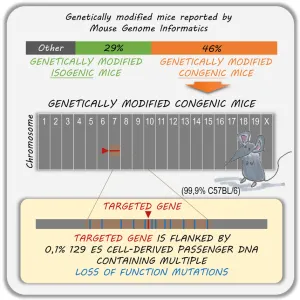
- Vanden Berghe, T., P. Hulpiau, L. Martens, R.E. Vandenbroucke, E. Van Wonterghem, S.W. Perry, I. Bruggeman, T. Divert, S.M. Choi, M. Vuylsteke, V.I. Shestopalov, C. Libert, and P. Vandenabeele. 2015. Passenger Mutations Confound Interpretation of All Genetically Modified Congenic Mice. Immunity. 43:200-209. (IF 20, 201 citations)
- All genetically modified congenic mice are populated with passenger mutations
- Phenotypic outcome of genetically modified congenic mice is potentially incorrect
- Phenotypic interference of passenger mutations is illustrated by some case studies
- A webtool provides a list of passenger mutations affecting genetically modified mice